Start selling in the EU
We act on behalf of your company as an Authorised Representative in the European Union and we help you with all the management and documentation necessary to Approval your CE Medical Product
Import medical device
Importing a medical device means bringing into the territory of the European Union (EU) a medical device that has been manufactured outside of it.
For example, if you buy medical products manufactured in Asia, the United States, or the United Kingdom, you are importing medical products.
To import medical devices into the European Union (EU) you need to meet a series of requirements; such as having a quality management system (QMS) based on ISO 13485, ensuring traceability, or collaborating with the Health Authorities when required.
In Spain, you also need the figure of the Technical Manager and the prior operating license (import activity) from the Spanish Agency for Medicines and Health Products.
Nosotros:
- We have experience in importing medical products,
- We are familiar with the typical errors in the medical device import process,
- We have mastered the applicable regulatory requirements and
- We have our own sanitary warehouse prepared to meet almost any requirement of the imported sanitary product.
If you need to import medical devices into the EU and you do not meet, or are unsure whether you meet, the applicable regulatory requirements, contact us!
What we offer?
MD Import offers you:
- Verification of the EU Declaration of Conformity, an essential document for the marketing of medical devices in Europe.
- Review of all the technical documentation of the medical device, to avoid incompatibilities, unforeseen events and unmet requirements.
- Representation before the European authorities of medical devices.
- Comply with the registration obligations applicable to medical devices in Europe.
- Cooperation with European authorities in the event of any required action or suspected adverse incident.
- Communicate and analyze customer complaints.
- Manage possible clinical investigations carried out in European territory.
- If necessary, we help you keep up to date the medical device documentation to ensure compliance throughout its life cycle.
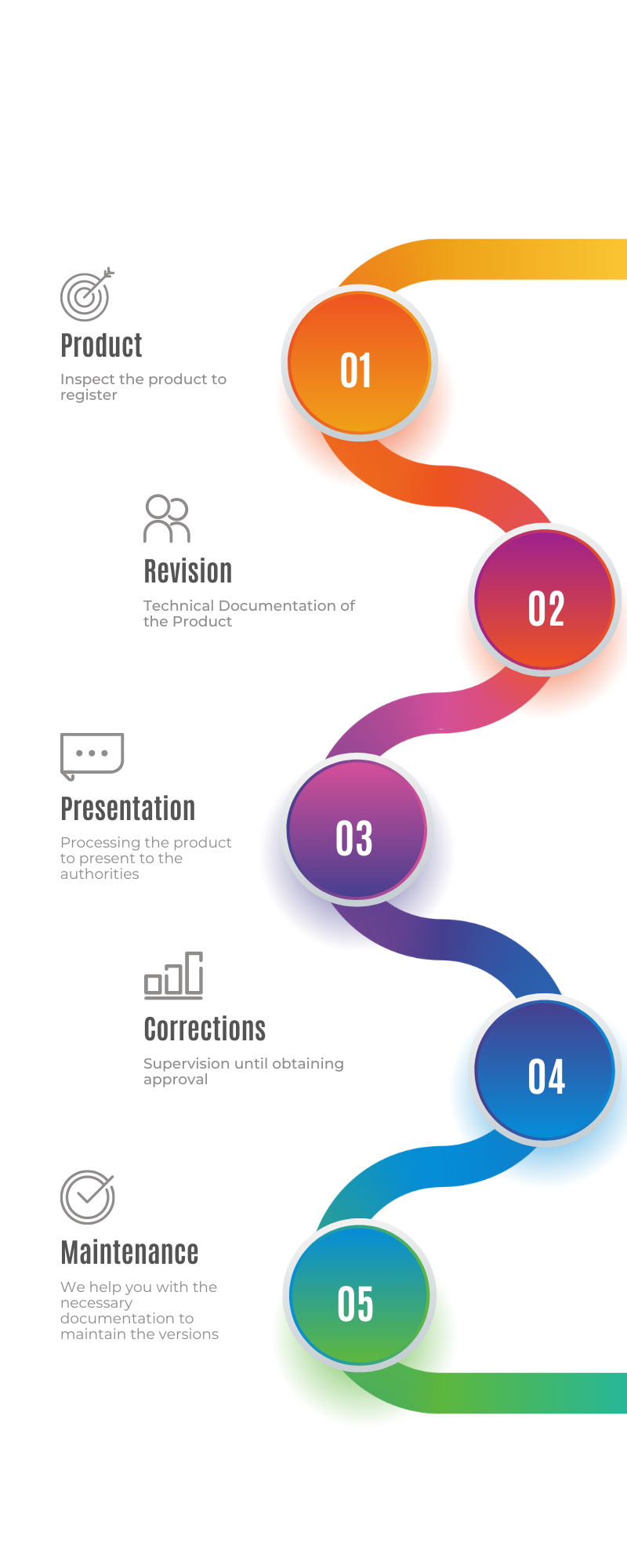
At MD Import, we manage the medical device obligations in Europe for your peace of mind. The responsibilities, commitments and obligations are specified in a written mandate that, signed by both parties, represents the essential framework for collaboration.
Who are we?
MD import
The experience and specialization of the MD Import technical team allows us to offer services in the field of import and regulatory compliance of medical devices in the European Union.
We have professionals with extensive experience in the medical device sector who routinely perform the responsibilities of economic actors in the medical device industry, particularly those of importers.
We are a multidisciplinary, serious and highly specialized team, which guarantees success in relations with medical device authorities and compliance with applicable requirements.
Who needs an Authorised Representative?
Companies that are not European and that want to sell their products within the European Union, either through eCommerce (electronic commerce) or physically.
Even if these products are sold through a supplier, it must be approved in the European Union.
An Authorised Representative is any person or company established in Europe that acts on behalf of a medical device manufacturer located outside the European Union. Therefore, it is a mandatory requirement for any medical device manufacturer located outside the European Union.
Why?
The role of the Authorised Representative (MD import) is mandatory for manufacturers of medical devices outside the European Union.
- Being represented by a company with experience and track record is a choice.
- Choosing your MD import to ensure that compliance with your (YOUR) obligations within the Union is handled efficiently and effectively is a choice.
- Hiring us as your MD import based on our track record, knowledge, and rigor is a smart choice.
As a foreign medical device manufacturer, the same general obligations apply as to companies located within the European Union, although distance, lack of experience with the new regulation and other aspects could make it difficult to market the medical product in Europe.
The marketing of the medical device in European territory could be at risk due to a lack of compliance. The new European Regulations (MDR and IVDR) are strict and demanding. You decide whether to take the risk or choose an authorised representative (MD import) that guarantees good results and has in-depth knowledge of the medical device and in-vitro medical device certification process.
